The Evaluation Process
NeuroDM was designed to remove the usual barriers to clinical evaluation. Our process guarantees high-level scientific analysis and economic validation in less than a month, thanks to agile governance and streamlined interactions with regulatory authorities (ANSM/CPP).
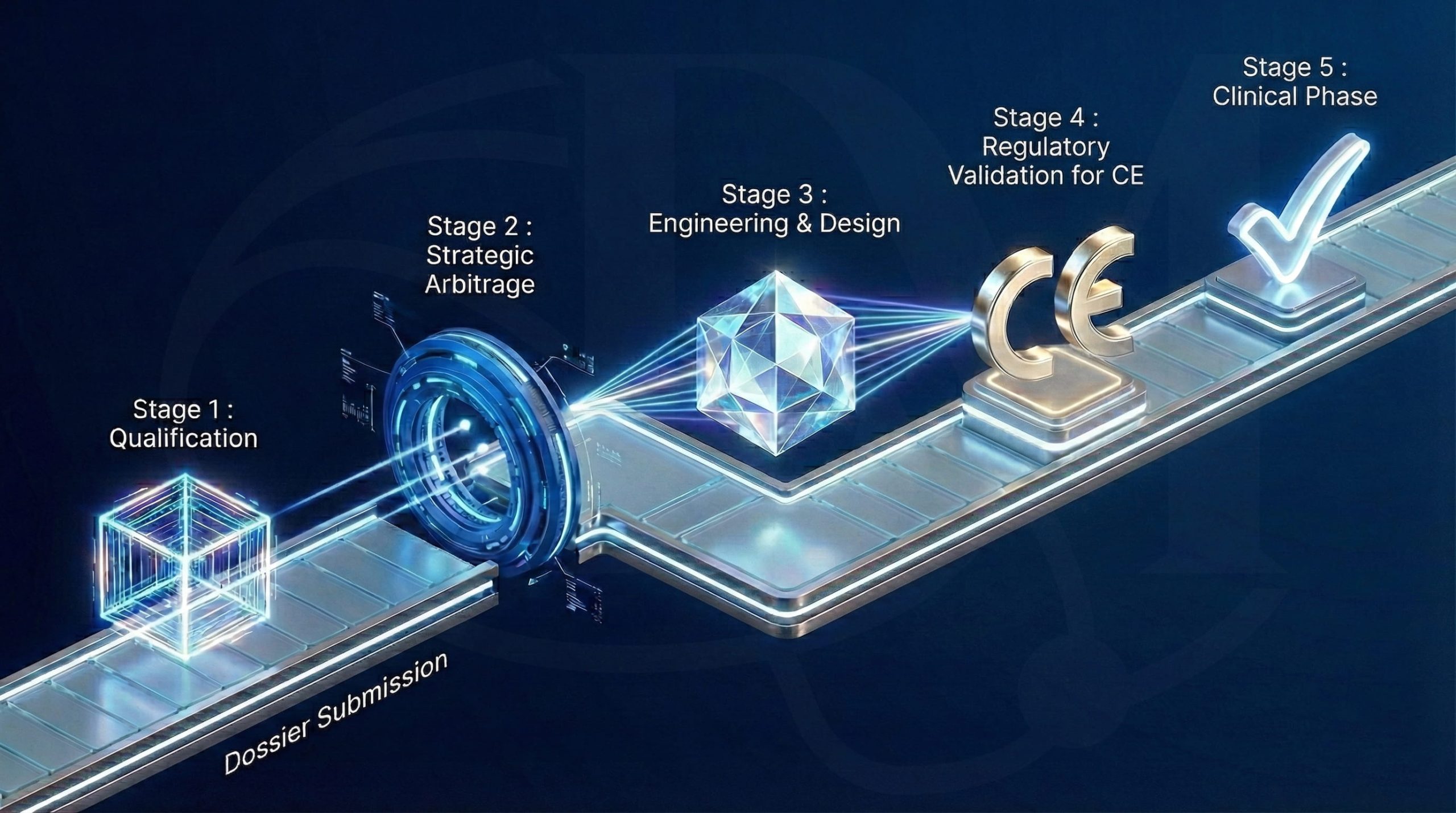
Your journey from idea to the operating room
The "Step-by-Step"
Qualification
Submission of your application via our online form.
Verification of completeness by R. Guizem

Strategic Arbitration
Presentation of your project to the Governance Committee
Evaluation of clinical interest and technical feasibility

Engineering & Design
Methodological scoping with the CEDM or the DRCI of the HFAR.
Budget establishment and signing of the agreement

Regulatory Go-Ahead
Submission to authorities (ANSM / CPP) by the DRCI Promotion Department or the DRCI of the HFAR.

Clinical Phase
Opening of centers, inclusions, and monitoring by our dedicated CRAs.
Our Added Value
NeuroDM does more than just evaluate your project; it provides an immediate boost to transform your innovation into clinical reality.
Accelerated Expertise
Parallel Engineering
Governance Responsiveness
Institutional Fast-Track
The NeuroDM Service Showcase
Your Investigation Catalog
Objectif: Marquage CE et Preuve de Concept
Études de Faisabilité (First-in-Human)
Investigations Cliniques Pivotales
Design de Protocole & Méthodologie
Soumissions Réglementaires :
Accès aux Plateaux Techniques
Objectif : Remboursement et Rayonnement
Suivi Clinique Après Commercialisation (PMCF)
Études de "Vraie Vie" (Real World Evidence)
Études Médico-Économiques :
Observatoires et Registres
Technical Pillars

Ready to save time?
Submit your application today for an initial response within 8 days.



